Published June 15, 2022
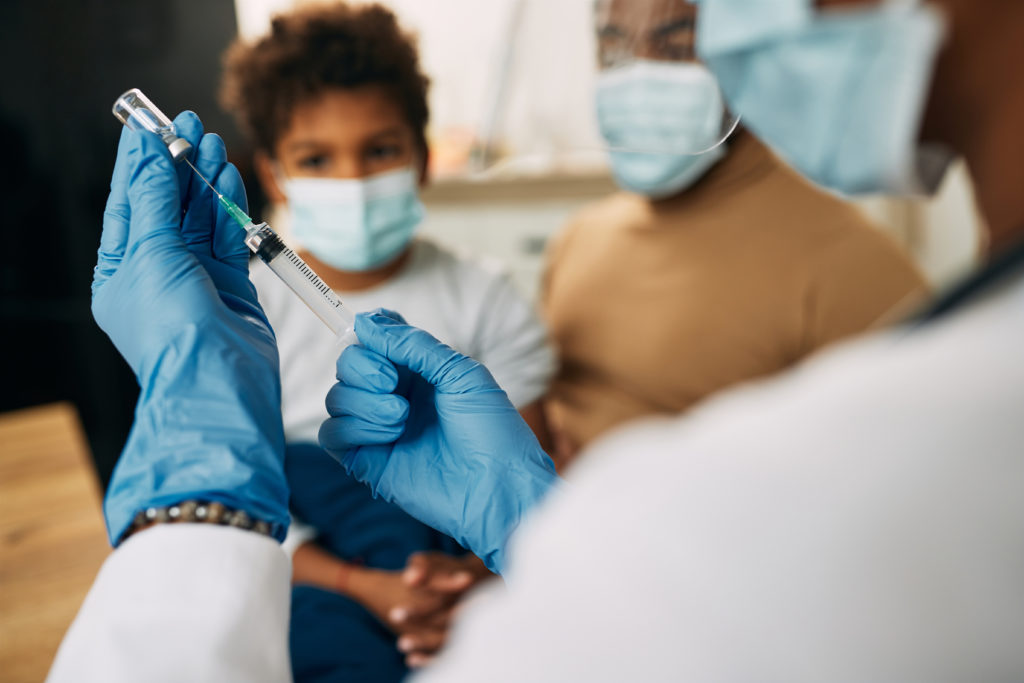
It won’t be long before many young parents find themselves in the doctor’s office, sitting with their little ones on their laps, faced with the all-important question: “Would you like little Johnny to get his COVID vaccine today?”
Upon inquiry of whether the vaccine is safe for their toddlers, many parents will likely hear, “Of course! It has now been approved by the FDA.”
In fact, such approval is set to happen this week as the FDA’s Vaccines and Related Biological Products Advisory Committee (VRBPAC) will meet to discuss recent requests to amend the Emergency Use Authorization (EUA) of the Moderna and Pfizer-BioNTech COVID-19 mRNA vaccines. Moderna is asking for its EUA to include the administration of the vaccine to infants, children, and adolescents six months through 17 years of age. Similarly, Pfizer-BioNTech is asking its vaccine EUA to include the administration of the vaccine to infants and children six months through four years of age.
But there is no need for a primary series of COVID-19 vaccines for children. The young have a decreased benefit from COVID-19 vaccination but are also at a greater safety risk. Plus, many children have already had COVID and have the benefits of naturally acquired immunity.
When deciding whether to approve these EUA requests there are several questions that VRBPAC must not ignore.
The first question is whether children who have naturally acquired immunity through previous infections should be vaccinated. This question is especially pertinent as a recent study sponsored by Moderna and Anthony Fauci’s National Institute of Allergy and Infectious Disease found that natural immunity is superior to immunity conveyed by any COVID-19 vaccine.
Since we do not have a full accounting of safety in adults, especially long-term risks, it seems inappropriate to propose mass vaccination in children, especially those who have already recovered from COVID.
The second question is whether vaccination of children is even needed at all.
We are no longer deeply embedded in the throes of the pandemic like we were in the summer of 2020. It is well-established that children, even without vaccination, have low risk of serious COVID complications, making it hard to justify vaccinating the younger age group.
The vaccines we are talking about were developed for the original strain of COVID-19, which has largely been replaced by newer variants such as Omicron. COVID-19 mutations are much less severe—a pattern that can occur with various viral mutations. There is also widespread availability of preventative, early-exposure, and early-treatment therapeutics with known safety records now, making the vaccines far less necessary.
Furthermore, 4 million vaccine doses must be administered to children ages five to 11 to prevent a single ICU admission, the Joint Committee on Vaccination and Immunisation has stated. Assuming two doses per child, that means two million children must risk potentially serious side effects to prevent a single child from requiring intensive care due to the Omicron variant of COVID-19.
These serious side effects are more widespread than most realize. Roughly 12 billion doses of the vaccine have been administered worldwide with over 825,000 reported COVID-19 vaccine adverse events reports in the U.S. alone. In all likelihood, this figure represents approximately 1 percent of the actual number of COVID-19 vaccine adverse events, according to a study from Harvard University’s Harvard Pilgrim Health Care Institute.

Of particular note, many of the more serious cardiovascular adverse events from COVID-19 vaccinations and boosters seem to disproportionately affect a younger population. Another independent analysis shows children under 18 are also 51 times more likely to die from the vaccine than a COVID infection if unvaccinated.
Despite the mild symptoms COVID brings to children and the high safety risk of the vaccines, the director of the FDA’s Center for Biologics Evaluation and Research, Dr. Peter Marks, stated he would not delay vaccine approval even if its efficacy was lower than the 50 percent threshold normally required in FDA guidelines.

Image: Screenshot from the vaccine approval meeting on June 15, 2022
It breaks all FDA norms and practices for the FDA to leap into an EUA so blindly and ignore decades-old standards of making careful safety- and efficacy-based decisions, especially when we are talking about our children. Leaving the bioethics argument and question of using our children as test subjects aside: Whatever happened to using hard clinical and scientific evidence as the basis for making decisions? What about the FDA and VRBPAC-employed physicians and their centuries-old sacrosanct vow of “doing no harm?”
The abandonment of the FDA’s decades-old efficacy and safety standards is part of the White House’s unrelenting push for COVID-19 vaccines to our kids no matter what. In fact, the White House has even plowed ahead as if approval was already in hand, announcing that vaccinations for young children could start as early as June 21st.
It is time for the FDA advisory committee members to stop blindly listening to the federal agencies, the White House, and mainstream news narratives for advice on clinical pharmacology and use their credentials, start their research from scratch, and make an unbiased review the safety, efficacy, and naturally acquired immunity data themselves. When it comes to the vaccine the data should be carefully examined and followed, not politics. Our children deserve no less.
Dr. David Gortler is a pharmacologist, pharmacist, and an FDA and health care policy oversight fellow and FDA reform advocate at the Ethics and Public Policy Center think tank in Washington, D.C. He was a professor of pharmacology and biotechnology at the Yale University School of Medicine, where he also served as a faculty appointee to the Yale University Bioethics Center. While at Yale, he was recruited by the FDA where he became a medical officer who was later appointed as senior advisor to the FDA commissioner for drug safety, FDA science policy, and FDA regulatory affairs. He is a columnist at Forbes, where he writes on drug safety, healthcare politics, and FDA policy.











