Published March 25, 2022
Properly evaluating any kind of drug safety — let alone that of presidentially mandated vaccines— involves carefully examining all of the available data. While Americans still haven’t been given full access to the full Pfizer “Comirnaty” license application, we do have access to the Food and Drug Administration’s medical officer review.
This comparatively spartan 107-page FDA review document purportedly contained everything the FDA believed was important to release to the public from the original Pfizer application of 329,000 pages. The FDA review conspicuously notes “clinically important serious adverse reactions [included] anaphylaxis and myocarditis/pericarditis”— that is, severe allergic reactions and inflammation of the heart and or the sac containing the heart, respectively.
Likewise, Moderna’s “Spikevax” vaccine was given its Emergency Use Authorization in late 2020, and was fully approved by the FDA on January 31, 2022. Following approximately a month of time to redact its review from “trade secrets,” the FDA released an even leaner 30-page review. Like the Pfizer vaccine, there were clear indications of myocarditis and pericarditis in the initial study, demonstrating the potential for risk going forward.
Specifically, the review states: “post-authorization safety surveillance has identified serious risks of myocarditis and pericarditis, particularly within 7 days following the second dose of Moderna COVID-19 Vaccine. The observed risk is higher in males under 40 years of age than among females and older males. The observed risk is highest in males 18 through 24 years of age… An increased risk for thrombosis with thrombocytopenia syndrome (TTS) has been identified following administration of adenovirus-vectored COVID-19 vaccines.”
Bottom line: The FDA, Centers for Disease Control, Pfizer, and Moderna knew about myocarditis and pericarditis risks from the mRNA vaccines since day one, yet made no formal warnings to Americans in the form of recommending labeling changes or any other warnings, such as letters to warn physicians, pharmacists, or the American public.
Drug Safety 101
Elementary drug safety education tells us that when any risk appears in small, preliminary, highly controlled, manufacturer-sponsored initial studies with specially selected subjects by the sponsor, there is significant potential that it would translate to many people having that risk when that same drug is administered to, say, much of the planet. In other words, it’s important to pay special attention to what adverse events manifest during the early stages of experimental drug testing, such as with the new mRNA technology.
Under the normal run of things, the responsibility of notifying consumers and prescribers of updates in safety and efficacy would fall upon manufacturers. But since Covid-19 vaccinations were mandated by the White House for all workers until the Supreme Court took action (they are still mandated for health-care professionals), and the fact that Pfizer and Moderna still have a federally granted liability shield, the duty of safety monitoring must fall under the auspices of the FDA.
Today, unquestionably serious cardiovascular, thrombotic, and neurologic adverse events related to the vaccine have occurred around the world. As I have written about before, the FDA’s own vaccine adverse event tracking system (the Vaccine Adverse Event Reporting System, or VAERS) shows substantial and serious risks from the vaccine, even though the FDA only collects an estimated 10 percent of all adverse events.
Still, federal agencies and manufacturers aren’t officially warning the American public about these risks, despite having been privy to this information for almost a whole year. Why? One reasonable explanation is because it would counter the narrative that endless vaccines and boosters is your patriotic duty.
It’s pretty clear today there are both safety and efficacy problems with vaccines and boosters. Because all of the FDA’s 18,000-plus employees had access to the same drug safety data we have, one must ask questions like these: Where is the updated Covid-19 labeling reflecting the latest safety and efficacy findings in VAERS?
Where are the FDA “Dear Doctor” letters giving updated safety guidance?
Where are the “Dear Pharmacist” letters to pharmacists who are still administering thousands of boosters to kids and other young healthy people on a daily basis?
Why isn’t the FDA recommending follow-up symptom tracking to avoid further inflammatory neurologic/thrombotic/cardiovascular tragedies instead of its proposal to extend the dosing interval and cross fingers that would mitigate risk (as there is no concrete clinical evidence that will happen)?
Why is the FDA ignoring internal drug safety epidemiologists who have stated during official FDA presentations that it only takes a single, well-documented adverse event to justify a safety signal investigation and warning to the American public of the risk?
Why isn’t the FDA demanding studies addressing genotoxicity, teratogenicity, oncogenicity, the potential for reduced fertility in men and women, the clinical effects of spike proteins in donated blood, and the bioaccumulation of vaccine in women’s ovaries? Why isn’t the agency convening and dedicating a Data Safety Monitoring Board to surveil all these post-marketing effects and others?
Are Americans expected to believe there is inadequate funding in the $6.5 billion-per-year taxpayer-funded FDA to address all of these public health issues?
Not Fully Disclosing Safety Risks Violates Standards
In order for a physician, pharmacist, and nurse, or anyone else with a clinical professional license to work at the FDA or CDC or any other public health agency, they must have a “current, active, full, and unrestricted license or registration from any state in the U.S.” Not fully warning patients about the potential dangers of any drug before administering useless and potentially dangerous vaccines and boosters would place these professionals’ license at risk, regardless of what the CDC, FDA, or White House says. Physicians, pharmacists, and nurses have always been expected to think for themselves rather than simply take orders.
As the truth is elucidated about vaccine efficacy and safety, these federal employees and mRNA vaccine manufacturers who colluded to withhold information from the public will be held accountable, and the whole “I was just following orders” excuse will not cut it. Just keeping your head down and cruising through your job is not an option when it comes to the lives of your fellow Americans. Licensures are held to a higher standard.
Once you see a rash of “early retirements” of federal public health employees (with full federal benefits, of course), expect starker evidence of clear malfeasance to come to light. When that happens, the licensed practitioners and scientists responsible for withholding vital health information from the public should be thoroughly investigated by their academic boards and licensing authorities.
Agency Missions Are Being Ignored
In addition, not speaking out appears to be a direct violation of the Federal Public Health Vision, Mission and Values, regarding its very specific obligations, and specifically to sections labeled “public health,” “accountability,” and “communication.” Federal public health employees’ silence also contradicts the FDA motto, which is to assure that: “All food is safe; all medical products are safe and effective; and the public health is advanced and protected.”
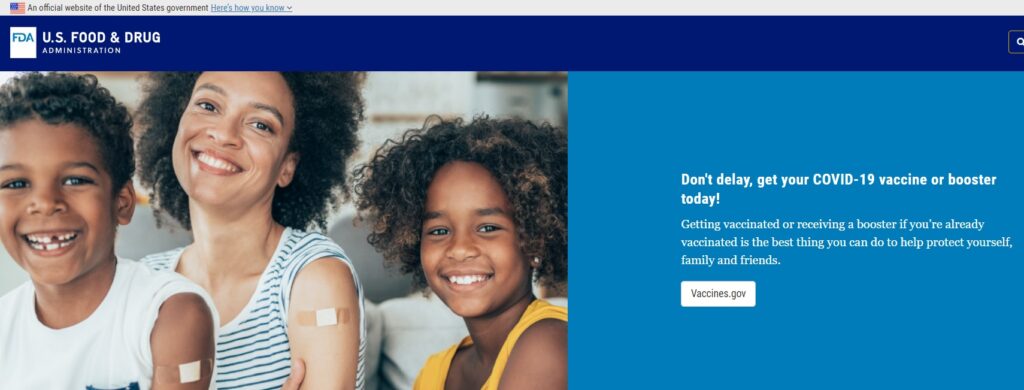
FDA officials are still pushing a potentially risky vaccine by purposely hiding facts from the public. Just look at today’s screenshot of the FDA website showing images of kids and young adults pushing boosters, despite the latest CDC data indicating there is no known benefit of Covid vaccination for those low-risk groups!
It will likely take a combination of courageous whistleblowers, a strong president who actually believes in “follow the science,” and an assertive new Congress to call the necessary hearings and issue the required subpoenas to uncover the many CDC and FDA civil and executive service malefactors who have taken the American people for fools.
These outrageously political, science-ignoring federal officials must be held accountable. Of course, nothing will happen to them while the Biden administration controls the White House and its pliant allies control Congress.
Even worse: If the Republicans gain control, will anyone other than Sen. Ron Johnson, R-Wisc., do anything to hold CDC and FDA officials accountable, or will they just again “reach across the aisle” and try to “find a middle ground” as they have done historically?
Dr. David Gortler is a pharmacologist, pharmacist, and an FDA and health policy fellow at the Ethics and Public Policy Center. He was a professor of pharmacology and biotechnology at the Yale University School of Medicine, where he also served at Yale’s Bioethicist Center, and was an FDA medical officer who was later appointed by the White House as senior advisor to the FDA commissioner for drug safety, FDA science policy, and FDA regulatory affairs. He is a columnist at Forbes, where he writes on drug safety, health care and FDA policy.
Photo by Mufid Majnun on Unsplash







